 The second figure serves as a visual aid for the table. The table of molecular geometries can be found in the first figure. A table of geometries using the VSEPR theory can facilitate drawing and understanding molecules. 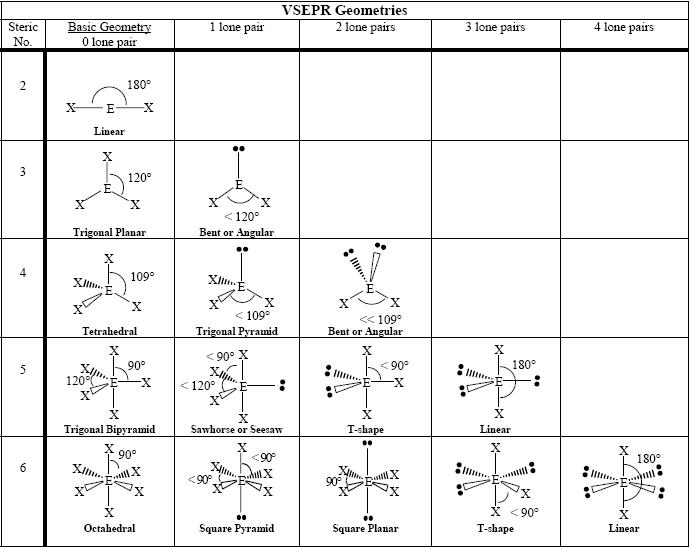 2]%2B%2B4.%2BTetrahedral.%2Bsp3.%2B[Cd(NH3)4]2%2B.jpg)
These structures can generally be predicted, when A is a nonmetal, using the "valence-shell electron-pair repulsion model (VSEPR) discussed in the next section. Molecular geometries (linear, trigonal, tetrahedral, trigonal bipyramidal, and octahedral) are determined by the VSEPR theory. It is only when there are one or more lone pairs on the central atom that molecular geometry differs from the electron pair geometry. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
